 
Explain how to obtain the conjugate acid of CO3^- base. 598 595 380 434 367 583 530 759 519 523 469 314 283 314 596 338 If H2O acts as an acid in a reaction, what would be its conjugate base? 704 690 717 717 717 717 717 596 717 682 682 682 682 603 580 600 Thus, for example, if the acid HF is compared with the base CH3COO, we expect the reaction to go part way to completion since the line is barely downhill. 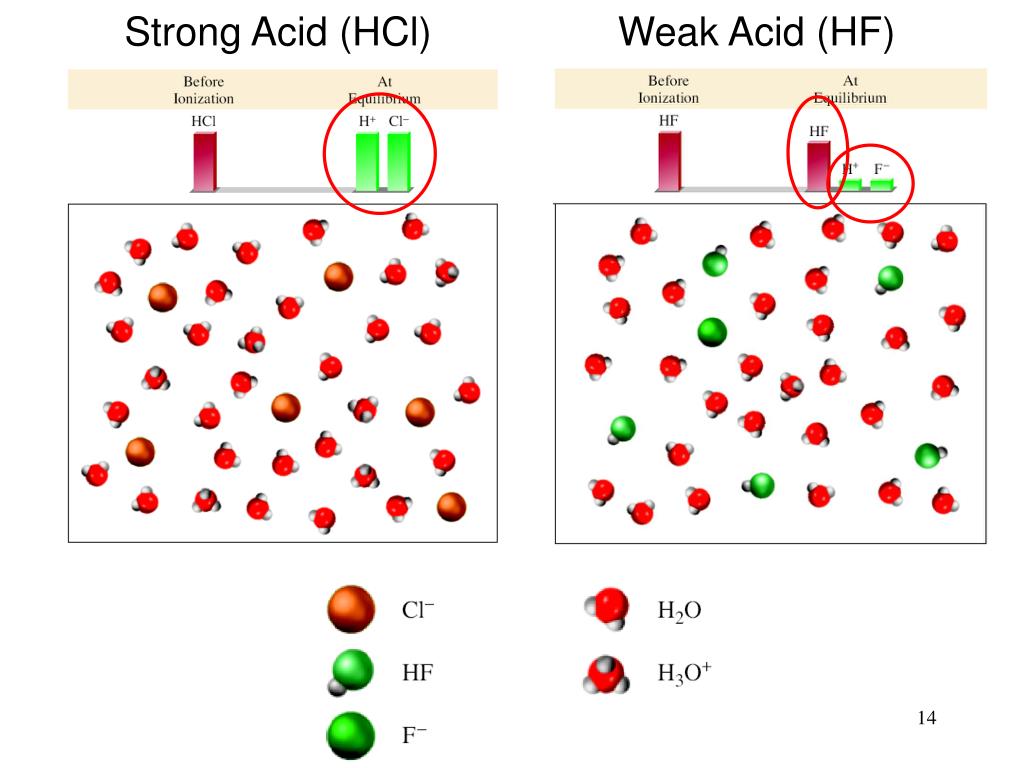
for communication, a whale song B) HSO_4^. She has taught science courses at the high school, college, and graduate levels. But NH3 is one of the compounds we know as a weak base. 
This problem has been solved! How do you determine the formula for the conjugate base of an acid? Replace immutable groups in compounds to avoid ambiguity. To book a personalized 1-on-1 tutoring session:Janine The Tutor proven OneClass Services you might be interested in:One. What are the conjugate acid and conjugate base of the bicarbonate ion, HCO3-? What is the conjugate acid in the following equation: NO2- +H2O->HNO2 + OH. Write the formula for the conjugate acid of the base NH2CH3. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. What is the conjugate base of the acid H2TeO3? KHSO4 + KMnO4 + H2SO4 = K2SO4 + MnSO4 + H2O. How To Balance C4H8ONH + H2O = OH using the algebraic method. Give the conjugate acid of the base ClO4. In your case, the base is hydrogen carbonate, or H CO 3. If we add one more proton or H+ ion to C4H8ONH we get, C4H8ONH2+. insufficient settled cash interactive brokers.exodus: gods and kings ending explained.willow oaks country club initiation fee.personification in wilderness by carl sandburg.engineering stress to true stress formula.bournemouth crematorium upcoming funerals.ohio state board of cosmetology laws and rules.is the cheytac intervention legal in california.humble apartments $400 a month all bills paid.je vous remercie pour votre aide et votre temps. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
